Learn how the structure of metal and the properties of different alloys impact common materials in jewelry creation. As a jeweler, there is nothing more rewarding than learning how to take your designs from paper to metal form.
When you are a beginner metalsmith, the design phase of jewelry making is a challenge. You may have an idea but you're not sure how to accurately put that on paper. As that wall breaks down, your designs will grow and develop. The next challenge will be with the metal as a material. You'll start to dream about geometrical shapes and folds, boxes and tubes. So, transforming ideas into three-dimensional jewelry will become your next challenge. Manipulating metal alloys for jewelry making can become a creative block if you don't understand the fundamentals of the structure including both the possibilities of the materials and the limitations.
In this article, we will explore the physical properties of metals for jewelry making. We will start with an overview of the molecular structure of elemental metals because this has a direct impact on the properties you will experience in the studio. Then, we will discuss what happens when you mix two or more metal elements to create an alloy. Most metals we work within the jewelry studio are alloys.
Jump ahead with these links:
Metal Structures
The different structures in pure metal are important in understanding how metals work. Odd as it may sound, metals have a crystal structure at the molecular level. It is an orderly construction with the electrons, which act as glue, binding everything together. This strong bond will keep the symmetrical structure in place. Three crystal structures, known as lattices, occur in pure metals.
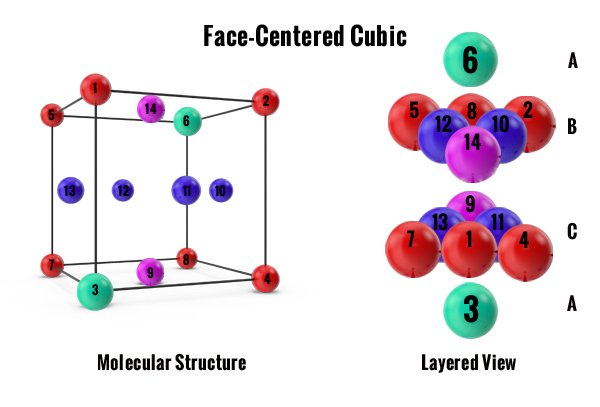
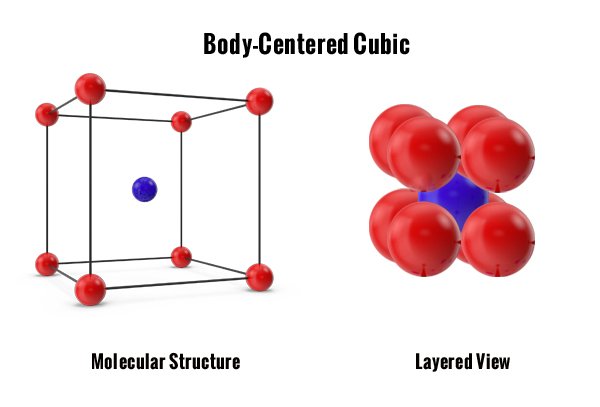
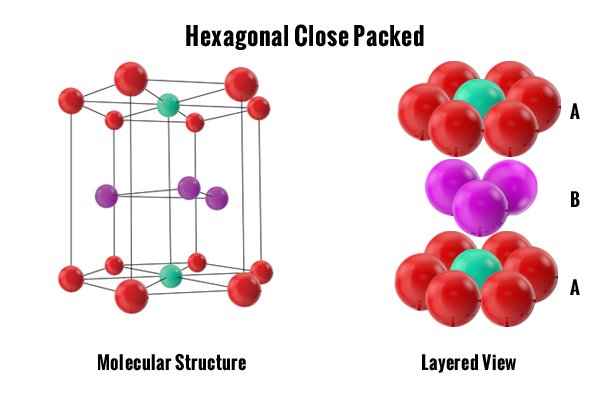
The first two are cubic structures. Atoms can slide over each other easily in these crystals because of the shape of the crystal cubes. The tightly packed FCC structure allows more slip planes than the loosely packed BCC structure. The total number of atoms inside of these cubic structures are important. The more atoms there are the more ductile the metal. The fewer atoms there are the stronger the metal. The odd one out is the HCP structure because it has 6 tightly packed atoms but with its hexagonal structure, there are fewer slip planes to allow ductility in the metal. Here is a brief example of each one.
Note: By looking at the structures below you can correlate by color where each atom sits in the molecular structure and then compare them to each layered view. The layered view is important in understanding how tightly packed the atoms sit together.
The most common molecular configuration in jewelry metals is the Face-Centered Cubic (FCC) crystal lattice structure. The cubic structure is tightly packed with 4 atom cells per unit as shown in the Molecular Structure graphic to the left. The structure is tightly shaped and the atoms easily glide over each other. Metals that share this structure are soft metals, referred to as ductile. They include gold, lead, nickel, aluminum, copper, silver, and platinum. These elements comprise the most common metal alloys for jewelry. Ductile metals can be shaped and drawn in the studio with tools.
The second arrangement, Body-Centered Cubic (BCC) structure, totals 2 atoms per unit cell. Packed cubic units build a symmetrical crystal structure. Because the structure is so loose and has fewer atoms, they do not move easily. This configuration makes it harder to manipulate the metal, creating a very strong material. Metals that have this structure are tungsten and alpha-iron.
Finally, the Hexagonal Close-Packed (HCP) structure is a tightly packed arrangement just like the FCC structure. However, there is one significant difference: the structure is hexagonal which stops the atoms from gliding over each other when force is applied. This structure is composed of 6 atoms per unit cell as shown in the graphic to the left. The metals that fall into this category are considered brittle because if you attempted to bend or form them by force they would break. HCP structured metals include zirconium, titanium, and zinc.
What is an Alloy?
Some pure metals, such as gold and silver, are too soft in their pure elemental state. Jewelers add different metals to strengthen the material and improve durability during wear. The resulting mix of two or more metal elements is called an alloy. The most common jewelry making metals are alloys: sterling silver sheet and 14 karat gold, for example. Base metals for jewelry making, such as brass sheet, are also alloys. Copper sheet is one of the few common materials that is near elemental purity.
Pure silver bends and scratches quite easily. A metal alloy for jewelry, like sterling silver, is a better solution for most applications. To make sterling silver you'd alloy 92.5% pure silver with 7.5% copper to have a stronger metal to work with. When both metal atoms are the same size they will become a substitution alloy, which means the new atoms will simply replace some of the base metal atoms in the structure. For example, silver and copper both have an FCC structure. When you add copper to the silver, the copper replaces some of the silver atoms. The FCC structure stays the same, the alloy retains its ductile properties and can be easily shaped or drawn in the studio.
If you have two metals combined that do not share the same atomic size, it becomes an interstitial alloy. An interstitial alloy changes the molecular structure of the main metal because the new atoms are too small to replace the atoms already there. These new smaller atoms that are introduced fall in between the larger original atoms to fill in the empty spaces. A perfect example is steel, which is an alloy of iron and carbon. The carbon atoms are much smaller than the iron atoms so they slip in between the iron atoms without replacing them.
Elements are alloyed to achieve different properties such as strength, malleability, durability, tarnish-resistance and sometimes just for color preferences. The resulting alloy maintains consistent properties throughout the entire batch of metal.
Note that layered materials are not alloys at all because they do not have uniform mixed elements throughout. Gold filled is an example of a layered material that is not an alloy.
What is Work Hardening in Jewelry Making?
The three metal structures, shown above, are nearly perfect when they are in their natural state. Once you start to apply force to the metal, whether hammering, piercing, cutting...etc, you are changing the original structure and so the metal becomes increasingly hard. The reason it becomes work hardened is that you are causing dislocations to occur in the crystal structures. When there are too many dislocations, the metal will eventually fracture. To restructure the crystals, you heat the metal at high temperatures then quench it or allow it to air cool (depending on the type of metal) in a process called annealing. You may need to anneal multiple times during metalwork to continue manipulating the metal. But, when a piece is complete, you may wish to keep it fairly work-hardened so the piece maintains its form during wear.
Another way to work harden metal is to heat it a certain way. For example, you can soften or harden sterling silver just by using heat. To soften sterling silver, heat it to 1200 degrees then let it air cool for 15 minutes then quench it. To harden it, heat it for 30-45 minutes at 600 degrees then allow it to air cool.
What is Annealing in Jewelry Making?
Halstead is one of North America's leading distributors of quality jewelry supplies. The firm is celebrating its 46th anniversary this year. Halstead specializes in wholesale findings, chain, and metals for jewelry artists.
Resources:
Wikipedia Alloys
NDT -Resource Center
ChemGuide
Total Materia
Annenberg Learner
Additional Articles:
9 Types Of Gold Metal Used In Jewelry
Halstead Lab Testing For Quality Control: A Jewelry Quality Assurance Program